 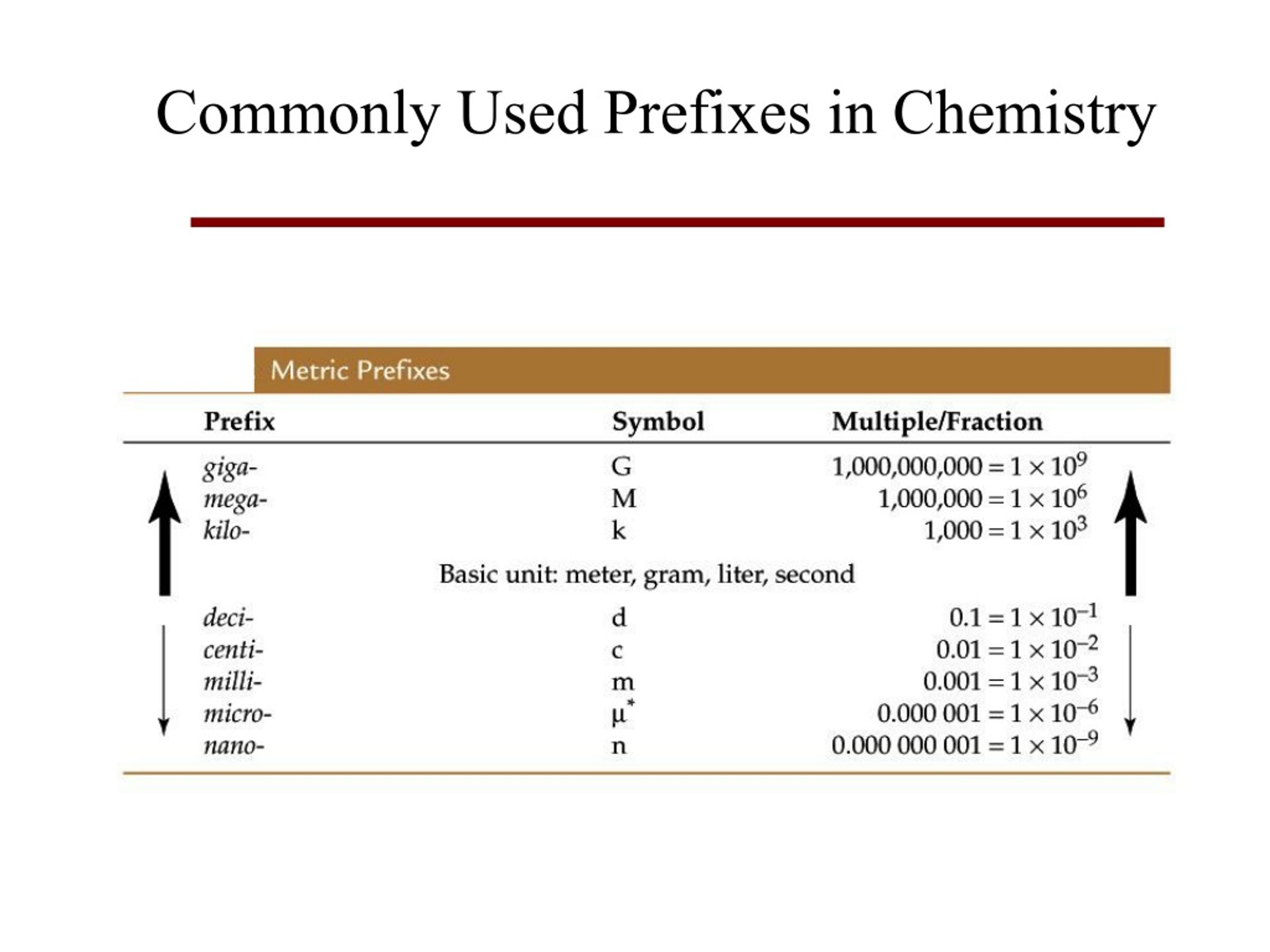
PRACTICE PROBLEMS: Give the name of each compound using a regular periodic tableand the covalent prefixes. Do not worry about the atomic number - remember, you will have a periodic table. How do you modify the ending of fluorine? Nomenclature is the naming of chemical elements and compounds. VIDEO Covalent (Molecular) Naming Demonstrated Example 1: Give the name of each compound using a regular periodic table and the covalent prefixes.Īnswer: It should be mono but since it is the first element you don’t write it. This rule is only true for the mono- prefix, so do not use it for any other purposes in covalent naming. It is a perfect example of how the first element, carbon, has no prefix, but the second has a prefix. So what does it look like? That is right: CO. 
Another common compound you have heard of is carbon monoxide. Here’s an additional example to drive the point home. If the first element in the compound is only one of that element, you do not use the mono- prefix. Ionic and molecular compounds are named using somewhat-different methods. So why does carbon not have a mono- prefix on it? The exception I was talking about earlier is just that. Chemists use nomenclature rules to clearly name compounds. Think… what is the name of it? Right, carbon dioxide. You have heard this exception before but may not have realized it. Mono- is a special prefix that you do not always use. One trick in this whole system has to do with the mono- prefix. Don’t worry about it too much, right now.Įxamples: Give the name of each compound using the covalent prefixes and a regular periodic table. Sometimes the “ a” on the end of the prefixes, like pent a or oct a, will be omitted. These prefixes are used in front of each elemental name in a compound. The prefixes names for the system are as follows: You describe the amount of each element by using a prefix word that indicates a number. How do you name covalent (molecular) compounds?įor the covalent system of naming, it depends on how many of each element there is in a compound. Examples of monatomic ions include Na +, Fe 3 +, Cl, and many, many others. This differentiates polyatomic ions from monatomic ions, which contain only one atom. The prefix poly- means many, so a polyatomic ion is an ion that contains more than one atom. 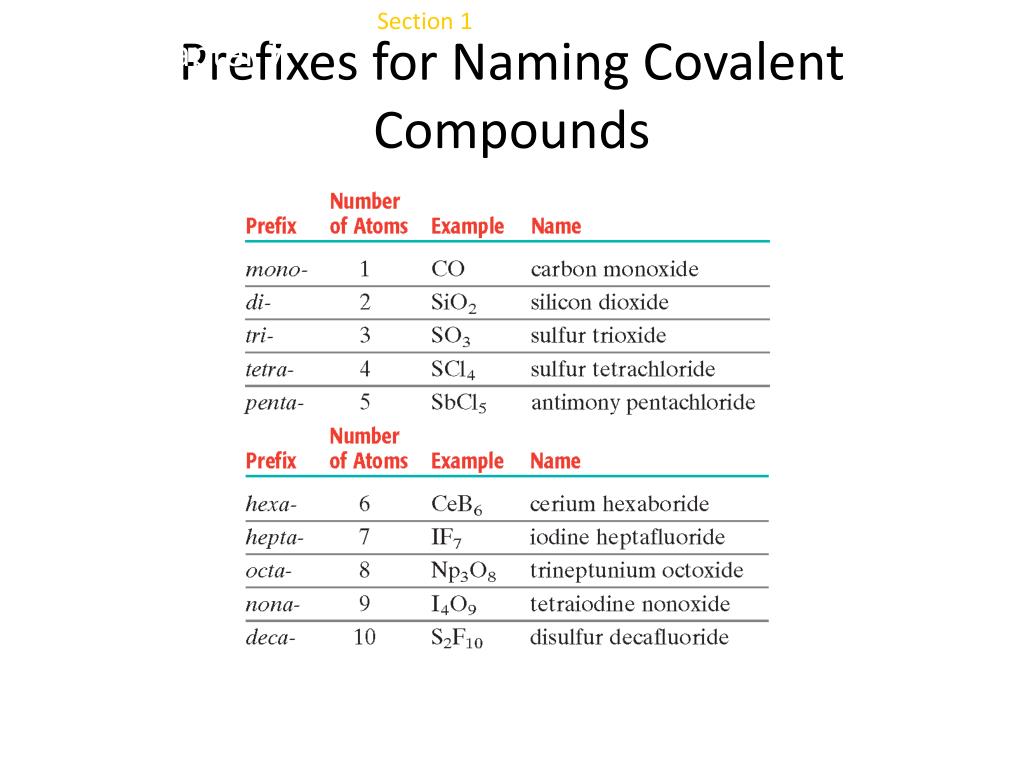
The four elements inside the group (P,S,As,Se), their names follow the same pattern but they have an extra oxygen atom.What sections should I know before attempting to learn this section? In this article, we will discuss polyatomic ions. Chemistry is a physical science, and it is the study of the properties of and interactions between matter and energy. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. You can find these charges in the periodic table below.Īs far as the naming goes, the group of elements (B,C,N,O,F, Br and I)will have naming pattern shown above for the chlorine family. Simulations - Discover a new way of learning Physics using Real World Simulations. Carbon will have 2-, and nitrogen will have 2. So, for example, the boron family of oxyanions will have a charge of 3. Remember, that in a family of oxyanions, the charge remains the same. Give the formula for: carbon dioxide, dinitrogen pentoxide. If there are two atoms, place the more 'metallic' first (furthest to the left on the periodic table) is there is a central atom, place it first, and the atoms attached after. Aerogels are transparent, low-density materials that are nearly 99. 2: Common greek prefixes used in naming simple moleculs. The small group of non-metals in the periodic table below commonly form bonds with oxygen, and they are also able to combine with oxygen in a number of ways to form different oxyanions. Report the diameter of the cell using the most appropriate prefix on the base unit of meter. Perchlorate – has the suffix “Per”, has the most number of oxygen atoms.(The prefixes are listed in Table 6.4.) TABLE 6.4. Hypochlorite – has the suffix “Hypo”, has the least number of oxygen atoms. They are named using prefixes to state how many atoms of an element are in one molecule of the compound.Look at the names of each polyatomic ions above. This group of oxyanions has a charge of 1.You will notice that the charge in a family of oxyanions remains the same.For example, chlorine can combine with oxygen in 4 ways to form 4 different oxyanions.Oxyanions are ions that contain oxygen atoms. Note: Chloride shown above is not a polyatomic ion, it's a monoatomic ion consisting of only one atom.Įxamples of other polyatomic ions: Oxyanions NaCl is sodium chloride, a combination of the name of the cation (sodium) and the anion (chloride). Do not use numerical prefixes if there is more than one ion necessary to balance the charges. Generally, polyatomic ions have suffixes that end in “ite” or “ate” if they contain oxygen atoms. Naming ionic compounds is simple: combine the name of the cation and the name of the anion, in both cases omitting the word ion. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
